CHOOSE TO
MOVE FORWARD
WITH STELARA®
Ready to take the next step toward symptom relief? Looking for a treatment option that could fit into your busy schedule? Your path to lasting remission could start when you talk to your doctor about STELARA®.

RAPID RELIEF
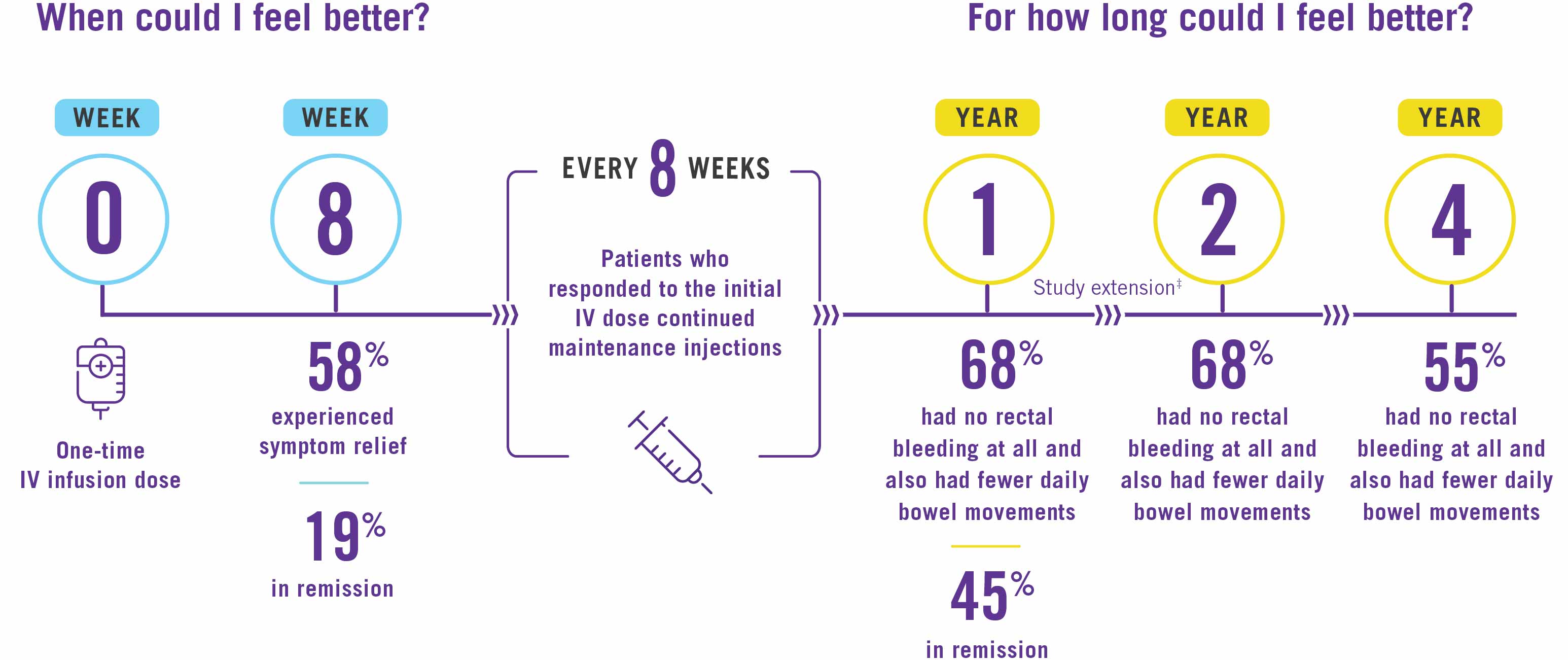
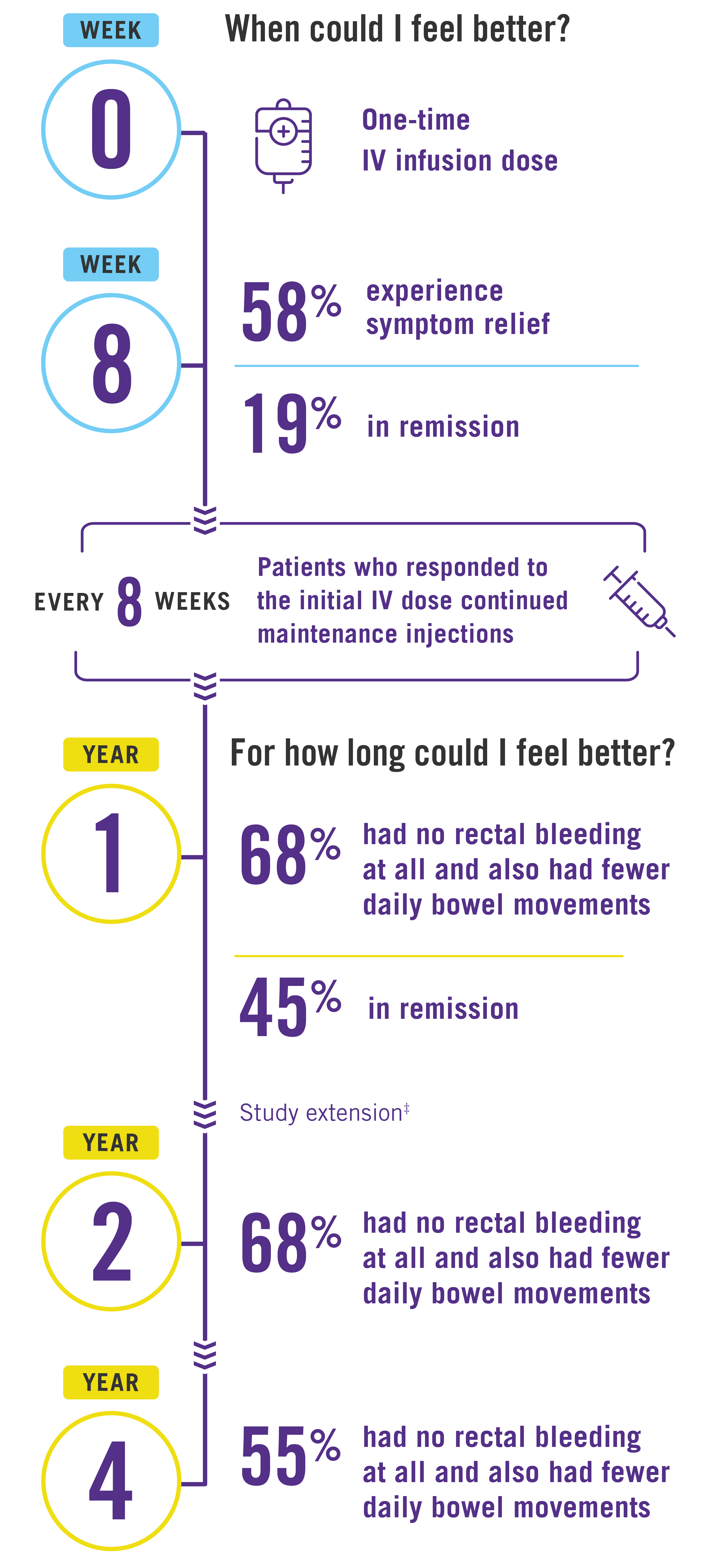
After only one intravenous (IV) infusion of STELARA®, the majority of patients saw relief from their UC symptoms in just 8 weeks, with nearly 1 in 5 achieving remission.*
LASTING REMISSION
4 out of 10 patients were in remission 1 year after responding to the IV induction dose and continuing treatment with STELARA®. The majority of patients had no rectal bleeding at all and also had fewer daily bowel movements at about 4 years.*†
Clinical trials of STELARA® included patients who failed or were intolerant to other medications, including a biologic, prior to STELARA®. Individual results may vary. STELARA® is not for everyone. Only you and your doctor can decide if STELARA® is right for you.
†After 1 year, patients and healthcare providers knew that STELARA® was being used. This may have increased results.
STELARA® RESULTS
‡After 1 year, patients and healthcare providers knew that STELARA® was being used. This may have increased results. Individual results may vary. STELARA® is not for everyone. Only you and your doctor can decide if STELARA® is right for you.
STELARA® IS THE FIRST APPROVED UC MEDICATION TO
REDUCE INFLAMMATION ON AND BELOW THE SURFACE OF THE INTESTINE.
Reduction of inflammation observed under a microscope and during colonoscopy in 17% of patients at 8 weeks and 44% of patients at 1 year.
This study did not evaluate how reduction of inflammation in the intestine relates to long-term outcomes.
LEARN ABOUT DOSING WITH STELARA®
Start with an IV infusion
ONE TIME
STELARA® treatment starts with a one-time IV infusion through a vein in your arm that provides the amount of medication based on your body weight and is administered by a medical professional in a comfortable setting. It takes at least 1 hour to receive the full dose of medicine.
Keep going with maintenance injections EVERY 2 MONTHS
After the one-time IV infusion, you will receive STELARA® as an injection under the skin (subcutaneous injection) every 8 weeks.
STELARA® is intended for use under the guidance and supervision of your doctor. If your doctor decides that you or a caregiver may give your injections of STELARA® at home, you should receive training on the right way to prepare and inject STELARA®.
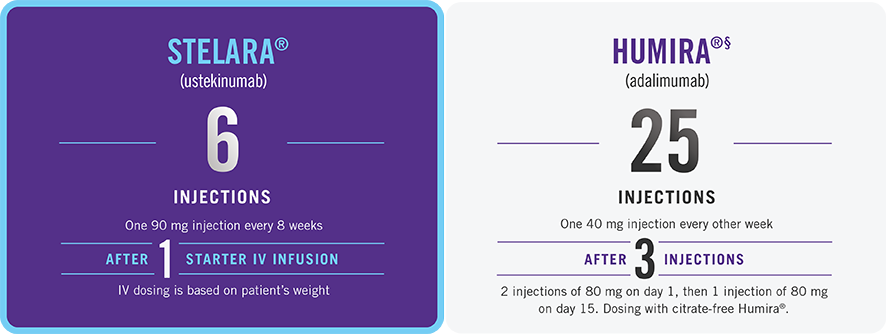
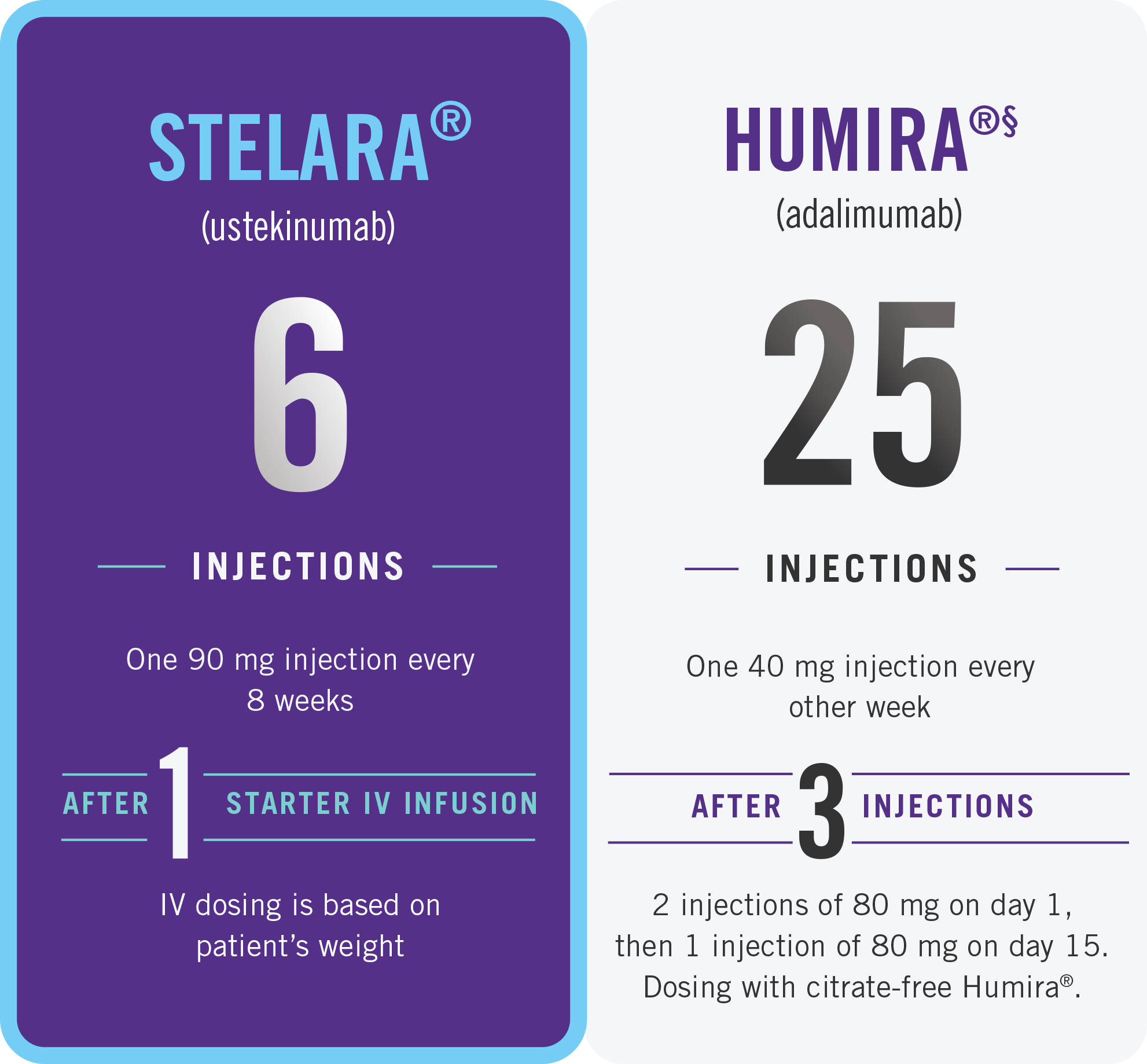
THE STELARA® DOSING DIFFERENCE
First 52 weeks of therapy after starter doses
While these factors are important, there are additional considerations for selecting treatment. Please talk to your doctor about treatment options and what might be right for you.
This presentation is not intended to compare the safety and effectiveness of these treatments. Please refer to each product’s full Prescribing Information for recommended dosing and administration.
§Indicated trademark is registered trademark of its respective owner.
THE LEGACY OF STELARA®
||This is based on information licensed from IQVIA™: LAAD claims, NPA, DDD for the period 2009-2021 reflecting estimates of real-world activity and based on custom methodologies developed by Janssen. All rights reserved.
¶The estimated number of patients treated with STELARA® in the United States is 22,000+ for UC and 121,000+ for CD.
MEET A STELARA® UC PATIENT
Transcripts:
" Where you are today is not where you have to spend the rest of your life. You have the ability to be the person that you were [before].
CHRIS
Actual Patient
What is STELARA® and how does it work?
- STELARA® is a prescription biologic medicine
- Many biologics are made from proteins, genes, or antibodies
- Some biologics target enzymes or proteins that may cause inflammation—like the inflammation thought to cause ulcerative colitis symptoms
STELARA® works differently
There are many different naturally occurring proteins in the body that contribute to inflammation. Two of these proteins, interleukin 12 (IL-12) and interleukin 23 (IL-23), have been implicated as important contributors to the chronic inflammation that is a main feature of ulcerative colitis. STELARA® is the only FDA-approved medication that targets these two proteins.
Transcripts:
STELARA® SAFETY
Ask your doctor about the benefits and risks of STELARA®. Prescription medications, including STELARA®, have possible risks involved with treatment, so it's important to discuss them with your doctor.
- nasal congestion, sore throat, and runny nose
- upper respiratory infections
- fever
- headache
- tiredness
- itching
- nausea and vomiting
- redness at the injection site
- vaginal yeast infections
- urinary tract infections
- sinus infection
- bronchitis
- diarrhea
- stomach pain
- joint pain
IS STELARA® RIGHT FOR YOU?
ASK YOUR DOCTOR TODAY.
When it comes to getting control of your UC symptoms, you are your own best advocate. The road to remission starts with an open and honest conversation with your doctor.

Transcripts:
" Your relationship with your doctor, your ability to communicate with one another, is as vital as your treatment plan itself.
STELARA® PATIENT BROCHURE
If you want to learn more about STELARA®, please download our patient brochure.
Once you and your doctor have decided STELARA® is right for you, STELARA withMe will help you find the resources
you may need to get started and stay on track. We will give you information on your insurance coverage, potential
out-of-pocket costs, and treatment support, and identify options that may help make your treatment more affordable.
STELARA withMe SAVINGS PROGRAM
- Eligible patients using commercial or private insurance can save on out-of-pocket medication costs for STELARA®
- Not valid for patients using Medicare, Medicaid, or other government-funded programs to pay for their medication
- Maximum program benefit per calendar year shall apply
- Terms expire at the end of each calendar year and may change
- Program does not cover the cost to give you your treatment
- See program requirements
STELARA withMe is limited to education about STELARA®, its administration, and/or the condition it treats. It is not intended to provide medical advice, replace a treatment plan you receive from your doctor or nurse, or serve as a reason for you to start or stay on treatment.